
It's a very good question. I really like it when N. asks questions like that. Technically, N. is doing science through his Kamana II studies, which is mainly the ecology of the Sandia Mountains, as well as related natural phenomena. But weather and climate are part of the local ecology.

The answer is not magic, it is sublimation. On earth, matter exists primarily in three states: solid, liquid, and gas, listed here in order of increasing energy. A fourth, and very energetic state of matter, plasma, is not so common on earth, but is very common in the universe.

So what is happening in the picture on the right, where there is no water--and, in fact, the temperature was 8 degrees F, which is well below the freezing point? Shouldn't the snow just hang around as a solid until the temperature gets high enough for a phase transition from solid to liquid?
In two words, not always.
Sometimes, when the vapor pressure at the surface of the solid is lower than the triple point for that substance, the whole liquid state is skipped. The state transits directly from a solid to a gas. This kind of phase change is called "sublimation."
Here, in our desert mountains, we lose a lot of snow to sublimation because the air is not capable of holding very much moisture due to altitude. Nor does it retain heat well, because of how dry it is.

In the picture, you may notice that the dirt now visible due to loss of snow from sublimation is frozen, and quite dry.
This continued very cold and clear weather due to a high pressure parked over the Four Corners region means that we will not get a lot of mud from melting of this snow cover.
And the 'shoe yekke' in me likes that. This means reduced vacuuming and mopping and washing of rugs.
On the other hand, this very common way for the snowcover to disappear also means that we do not retain as much water in the soil, perpetuating the dryness of our desert mountains.
Science:It's everywhere!
Even on our morning walk.














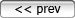




2 comments:
That is so neat! I don't think any of our snow sublimated, though, judging by Bo's muddy paws.
I love hearing those questions too. It is fun to chat and work through some logical possibilities before getting to the known facts. Fun!
Post a Comment